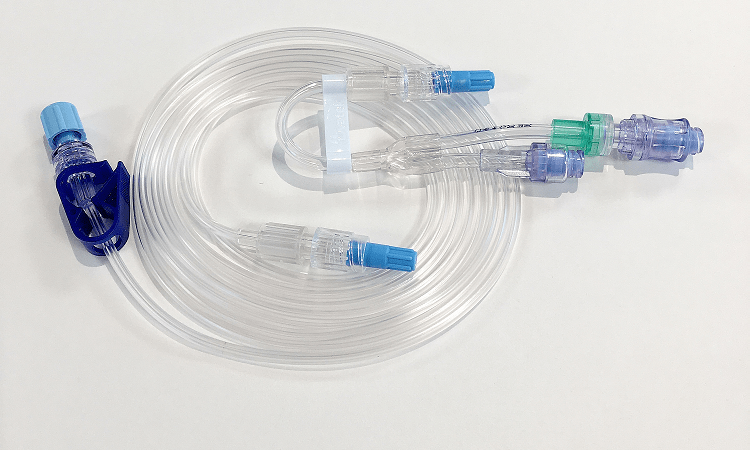
State-of-the-Art Medical Flow Sensor for Respiratory Monitoring
Best practice clinical protocols advise that neonatal ventilation flow sensors should use both lung-protective volume control and CO2 monitoring. Clinicians who do not combine volume and CO2 monitoring in the smallest patients, tend to do so because the excessive dead space will not let them.
The Europlaz Flow Sensor is a patented neonatal ventilator flow sensor that enables clinicians to follow the correct protocols, by combining the sensor for measuring flow volume with an optimally placed sampling port for safely extracting gas for tidal flow CO2 monitoring.
This advanced combination medical flow sensor ensures you get accurate readings and as a result, are in a better position to monitor an infant’s condition and recommend treatment.
Europlaz is a cutting-edge medical technology manufacturer. Our patented ventilation flow sensor is produced using automated micro-robotic assembly, for a highly consistent sensor quality, performance and traceability.
For more information on our medical flow sensor please download the brochure and free whitepaper, call +44 (0)1621 773 471 or email [email protected].
Europlaz’s Medical Flow Sensor
Key information:
- Integrated sidestream CO2 sampling port
- Lowest in class 0.8ml dead space
- Lowest in class 7.5g weight is at least 50% lighter
- Mistake-proof circuit assembly. One less connector reduces disconnect risk by at least 30%
- Secures a true measurement and completely eliminates estimation errors in lung protective volume ventilation
- Lower gas turbulences assuring measurements accuracy
- Reduced net purchasing and stocking cost
Our Flow Sensor is produced using automated micro-robotic assembly, for a highly consistent sensor quality, performance and traceability.
Single-use and reusable versions are available with high supply capacity for assurance of timely delivery. Customised branding and packaging are also available.
For further information please see our Flow Sensor data sheet.


Other Flow Sensors on the Market
Key information:
- Combined deadspace of 2ml to 5ml
- Large, unyielding and uncomfortable
- Prone to connectors mismatch and human errors, which can result in significant measurement errors
- Elevated risks of lung protective volume errors, from ventilator sample gas estimation and connection errors or secretion in sampling tube
- Higher gas turbulences contributing to flow and CO2 measurement errors
- Cost of two separately stocked parts
Comparison to Historic Ventilator Flow Sensors
In sidestream CO2 monitoring, a chain of connectors can by nature not entirely mitigate human errors, such as not pushing them fully together or inadvertently placing them in the wrong order.
Placing the side-stream adapter before the flow sensor, for example, will result in a significant tidal volume error, because the ventilator assumes post-sensor sampling gas loss that does in fact not occur.
Worst case, the ventilator overshoots tidal volume by 50%. Also, the turbulent gas mixing will result in an EtCO2 under-measurement.


Risks to Neonatal Patients
The smallest patients require a minute volume as low as 100ml. When considering that the sidestream adapter draws 50ml/min, this means that the medical flow sensor must estimate and compensate for 50% of the tidal volume.
Any secretion and sampling line obstruction, or sidestream pump wear, has the potential to create a significant estimation error and volume inaccuracy. Such an error will in turn affect the calculation of compliance and resistance values; potentially providing incorrect feedback to clinicians. This carries a safety risk in lung-protective ventilation of the smallest and most fragile patients.
Additionally, dead space from the extra connector can be a significant detractor for use with the smallest babies. So-called ‘low dead space’ adapters still add a significant volume, where the smallest patients are concerned.
This is a concern when it comes to the common design convention of using a flexible, contracting bore reducer as it can risk a slight misalignment to the small ET connector bore, with undesirable flow resistance and turbulence (measurement noise) results.
Benefits of Europlaz’s Advanced Combination Medical Flow Sensor
The Europlaz combination flow sensor eliminates all of the concerns in existing neonatal ventilation flow sensors. The patented position and distance between the side-port orifice and the anemometer flow sensor is constructed to eliminate inaccuracies from the gas turbulence noise that can occur between multiple connectors ensuring accurate results unaffected by noise such as resistance or turbulence.


Compatible Machines
You can contact your ventilator supplier to enquire about their variant of the Europlaz medical flow sensor with a combined CO2 sampling port.
Infant ventilators for which the Europlaz Flow Sensor is 100% compatible with:
- Acutronic Florian Respiratory Monitor
- Acutronic Fabien NCPAP Evolution
- Acutronic Fabien HFO
- Acutronic Fabien HFOi
- GE Engestom Carestation Neonatal
- Heinen & Lowenstein Leoni plus
- Maquet Servo-N
- SLE 2100 VPM
- SLE4000
- SLE5000
- SLE6000
Infant ventilators using hotwire technology for which the Europlaz flow sensor is not 100% compatible but could potentially be adapted with sideport technology include:
- Draeger Babylog8000
- Draeger VN800
- Vyaire Avea