Contract Manufacturing of Drug Delivery Devices
With specialised capabilities for combination products, our end-to-end network provides the comprehensive expertise you need to launch a commercially viable product.
Experience in Drug Delivery Devices
Our experience lies with a range of drug delivery technologies, including wearable and non-wearable auto-injectors, IV sets and connectors, respiratory devices, and more. With services ranging from development to moulding, assembly, and validation, we provide a complete service from start to finish.
These capabilities allow the Europlaz team to develop and manufacture devices which are ideally suited to support the needs of your active ingredient. We manufacture hundreds of millions of commercially successful devices each year – quick, cost-efficient whilst of the highest quality.
We are highly skilled in developing complex drug delivery devices for mass manufacturing. One of our largest clients is a global, blue chip healthcare manufacturer, where we have previously supported them in manufacturing 53 million components per year for their inhalers.
This experience along with our contract manufacturing capabilities allows us to combine services, technologies and products into tailored drug delivery solutions to improve patients’ lives. All the while delivering integrated solutions from early development of drug delivery systems to finished combination products.

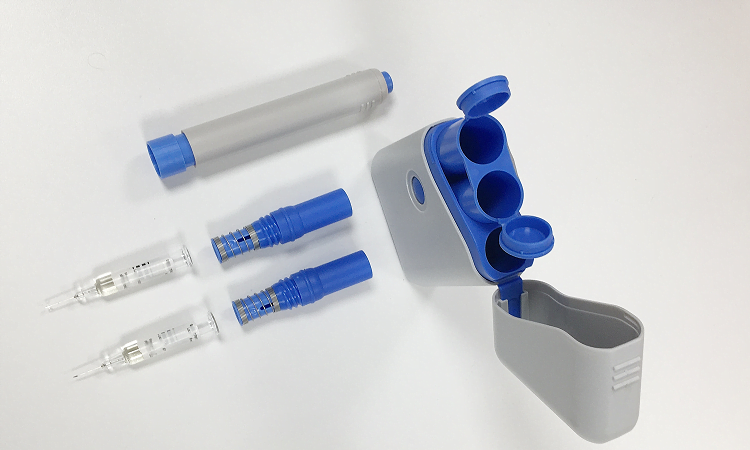
Bringing your Device to Market
Understanding and following the regulatory processes of a drug delivery device is often challenging. By working with Europlaz, you will have experts on hand to guide you through these often-complex regulations. This specialist industry knowledge will support you in getting your product to market faster.
Manufacturing Excellence for Healthcare Innovation.
Helping OEMs and Inventors to scale their businesses by partnering with a flexible medical device contract manufacturer.
Want to find out more?
Types of Drug Delivery Devices
Drug delivery devices are specialised tools and technologies designed to administer medications or therapeutic substances to patients. These devices are essential for delivering drugs in a controlled and efficient manner. They come in various forms to suit different drug types, administration routes, and patient needs.
Much of our work in the drug delivery market falls under confidentially agreements.
However, opposite are some common categories of drug delivery devices that we can help manufacture components and parts for. Scroll through the images for a complete list.
To find out more about our medical device contract manufacturing services please contact us today.

Download our brochure
Curious to learn more? Download our brand-new company brochure to discover how Europlaz is evolving and what it means to you.
Discover Europlaz
Our UK-based ISO 13485 certified and FDA-compliant cacility means that we have complete control of the process, from under one roof. We are flexible, and resourceful, and can take on projects of any size. Our clients range from small start-ups to international blue chip medical device suppliers.
Book a Meeting
To learn more about Europlaz’s services and the different medical devices we have helped develop, book a telephone meeting with one of the team.
To do this simply use the Schedule Meeting button below.
Alternatively you can contact us by telephone or email.
